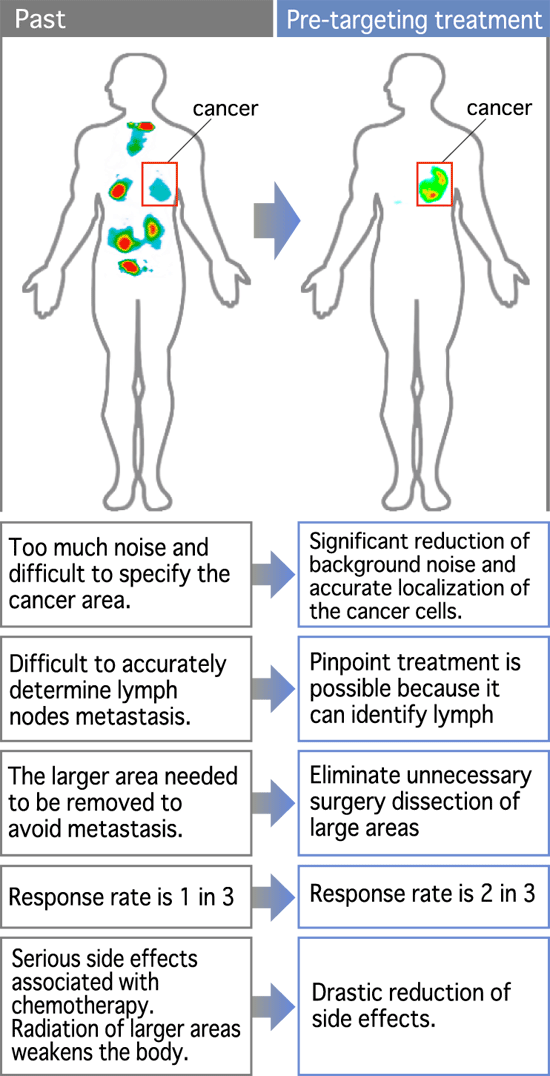
Our pre-targeting method pinpoints target cancer cells through imaging using high affinity monoclonal antibodies.
First, this method provides us accurate images of cancer cells in whole body. This will make it possible for clinical doctors to have many options for the most effective treatment of cancer with the least pain and side effects.
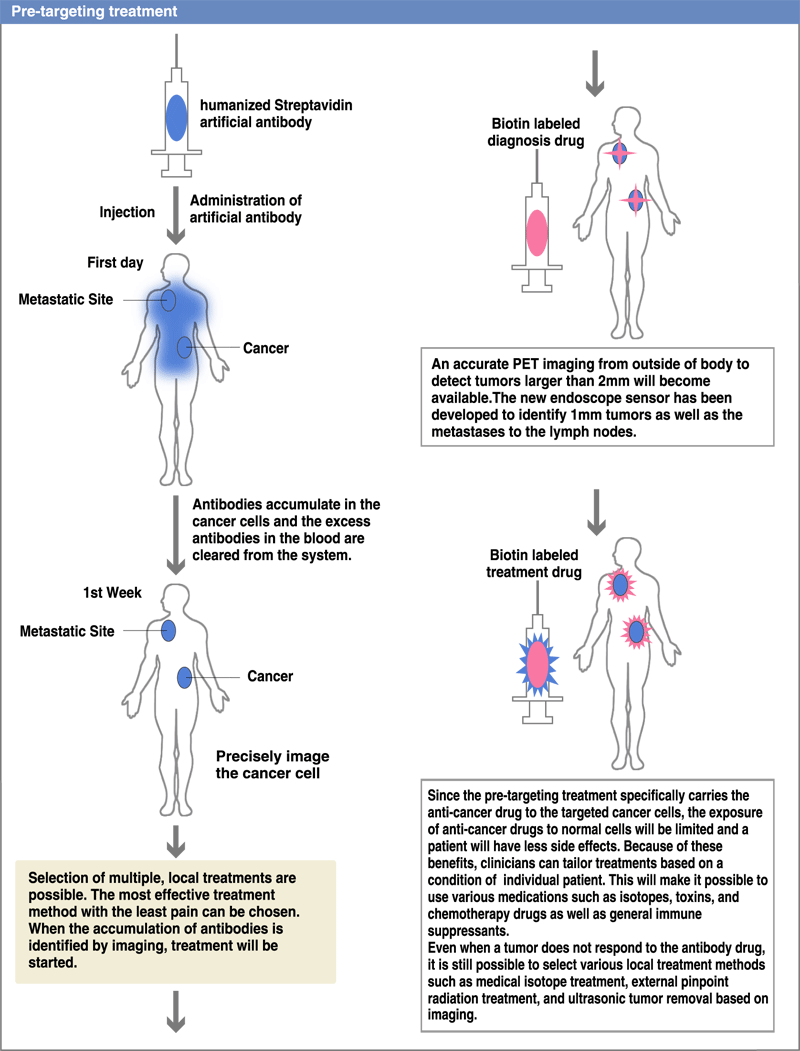
Since the pre-targeting treatment can specifically deliver anti-cancer drugs to the antigen-expressing cancer cells, the exposure of anti-cancer drugs to normal cells will be limited and the patient will have fewer side effects. Because of these benefits, clinicians will be able to tailor treatments for an individual patient with the use of various medications such as isotopes, toxins, or anti-cancer drugs as well as immune suppressants based on his or her condition. (Diagram 1)
First, this method provides us accurate images of cancer cells in whole body. This will make it possible for clinical doctors to have many options for the most effective treatment of cancer with the least pain and side effects.
Since the pre-targeting treatment can specifically deliver anti-cancer drugs to the antigen-expressing cancer cells, the exposure of anti-cancer drugs to normal cells will be limited and the patient will have fewer side effects. Because of these benefits, clinicians will be able to tailor treatments for an individual patient with the use of various medications such as isotopes, toxins, or anti-cancer drugs as well as immune suppressants based on his or her condition. (Diagram 1)
significantly reduce side effects of cancer treatment
PET (Positron Emission Tomography) is widely used for cancer diagnosis based on it high sensitivity and accuracy to localize cancer cells. However, accumulation of isotope in brain and other organs increase the background level which results in low specific resolution.
In contrast, immune-PET identifies cancer cells using highly specific antibodies with the high accumulations of isotopes in cancer cells and low background. However, these antibodies have high molecular weight and patient wil be exposed to excessive radiation if the radioisotope is directly coupled to the antibody molecules.
In our pre-targeting method, we will administer non-label antibodies first. After one to three weeks, the target antibodies will accumulate in the cancer cells and the excessive antibodies will be flushed out from the body. We will then administer low molecular weight radioactive compound. The unbound compounds will be cleared quickly from patients through the kidneys. Thus, non specific radiation exposure will be kept to a minimum level. When using this method, the antibodies used in the first step and the small molecules used in the second step will have to bind with high affinity to form stable complex. To achieve this goal, we will use Streptavidin (SA) as the antibody and biotin as the labeling compound. (Diagram 2)
In order to make this method successful, it is critically important to ensure that the affinity of the antibody is high enough for the target molecule on cancer cells so that the antibody-target complex will remain stable during the treatment. For this purpose, we will artificially change the antibody to meet this specification using computer simulation design.
Based on our past experience in creating genome-antibody drugs, researchers at MDADD have been successful in producing antibody imaging PET, purification of Cu64 and developing a highly sensitive endoscope sensor are some of the examples.
Currently, we can identify tumors as small as 3 mm diameter. We are currently trying to identify 2mm tumors from outside of body. (In an associated project, we are in the process of developing a device that will be used with an endoscope. The device will detect positrons in close proximity). Our goal is to identify cancer cells using the Pre-targeting method, lowering the noise level and raising signal sensitivity. Ultimately we aim to identify 1 mm tumors. This will make us possible to accurately diagnose metastasis in the lymph nodes.
In contrast, immune-PET identifies cancer cells using highly specific antibodies with the high accumulations of isotopes in cancer cells and low background. However, these antibodies have high molecular weight and patient wil be exposed to excessive radiation if the radioisotope is directly coupled to the antibody molecules.
In our pre-targeting method, we will administer non-label antibodies first. After one to three weeks, the target antibodies will accumulate in the cancer cells and the excessive antibodies will be flushed out from the body. We will then administer low molecular weight radioactive compound. The unbound compounds will be cleared quickly from patients through the kidneys. Thus, non specific radiation exposure will be kept to a minimum level. When using this method, the antibodies used in the first step and the small molecules used in the second step will have to bind with high affinity to form stable complex. To achieve this goal, we will use Streptavidin (SA) as the antibody and biotin as the labeling compound. (Diagram 2)
In order to make this method successful, it is critically important to ensure that the affinity of the antibody is high enough for the target molecule on cancer cells so that the antibody-target complex will remain stable during the treatment. For this purpose, we will artificially change the antibody to meet this specification using computer simulation design.
Based on our past experience in creating genome-antibody drugs, researchers at MDADD have been successful in producing antibody imaging PET, purification of Cu64 and developing a highly sensitive endoscope sensor are some of the examples.
Currently, we can identify tumors as small as 3 mm diameter. We are currently trying to identify 2mm tumors from outside of body. (In an associated project, we are in the process of developing a device that will be used with an endoscope. The device will detect positrons in close proximity). Our goal is to identify cancer cells using the Pre-targeting method, lowering the noise level and raising signal sensitivity. Ultimately we aim to identify 1 mm tumors. This will make us possible to accurately diagnose metastasis in the lymph nodes.